Biological Molecules - Organic Chemistry
Card 1 of 176
Chemists and biochemists have many ways of representing sugars. Glucose, the most common hexose, is shown below in various linear and cyclic projections. Using the linear and cyclic projection of your choice, can you indicate which colored oxygen in the linear form corresponds to the circled hemiacetal oxygen once the cyclization reaction is complete?
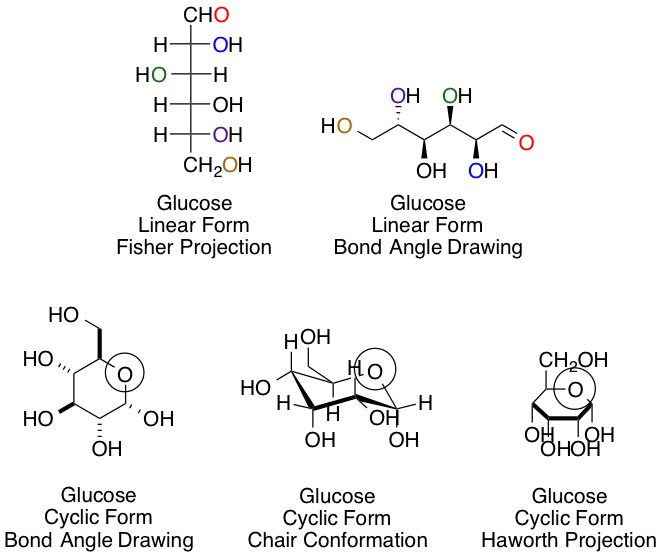
Chemists and biochemists have many ways of representing sugars. Glucose, the most common hexose, is shown below in various linear and cyclic projections. Using the linear and cyclic projection of your choice, can you indicate which colored oxygen in the linear form corresponds to the circled hemiacetal oxygen once the cyclization reaction is complete?

Tap to reveal answer
This answer, regardless of your preference of projection type, is easiest to obtain using arrow pushing for the cyclization reaction to keep track of each carbon and oxygen:

The purple carbon in the linear projection ends in the circled hemiacetal position.
This answer, regardless of your preference of projection type, is easiest to obtain using arrow pushing for the cyclization reaction to keep track of each carbon and oxygen:

The purple carbon in the linear projection ends in the circled hemiacetal position.
← Didn't Know|Knew It →
Which of the following structures represents the anomeric alpha ring structure of D-glucose?

Which of the following structures represents the anomeric alpha ring structure of D-glucose?

Tap to reveal answer
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the  attached to carbon 5, while those that are in the left position end up cis to the
attached to carbon 5, while those that are in the left position end up cis to the  attached to carbon 5.
attached to carbon 5.
If the hydroxyl group attached to carbon 1 ends up trans to the  attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the
attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the  attached to carbon 5, the ring structure is considered beta.
attached to carbon 5, the ring structure is considered beta.
The alpha ring structure of D-glucose bonds the carbon 1 hydroxyl group trans to the carbon 5  group. The hyroxyl groups on carbons 2, 3, and 4 will be trans, cis, and trans with respect to the
group. The hyroxyl groups on carbons 2, 3, and 4 will be trans, cis, and trans with respect to the  .
.
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the 

If the hydroxyl group attached to carbon 1 ends up trans to the 

The alpha ring structure of D-glucose bonds the carbon 1 hydroxyl group trans to the carbon 5 

← Didn't Know|Knew It →
Which of the following structures represents the anomeric alpha ring structure of D-galactose?

Which of the following structures represents the anomeric alpha ring structure of D-galactose?

Tap to reveal answer
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the  attached to carbon 5, while those that are in the left position end up cis to the
attached to carbon 5, while those that are in the left position end up cis to the  attached to carbon 5.
attached to carbon 5.
If the hydroxyl group attached to carbon 1 ends up trans to the  attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the
attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the  attached to carbon 5, the ring structure is considered beta.
attached to carbon 5, the ring structure is considered beta.
The alpha ring structure of D-galactose bonds the carbon 1 hydroxyl group trans to the carbon 5  group. The hyroxyl groups on carbons 2, 3, and 4 will be trans, cis, and cis with respect to the
group. The hyroxyl groups on carbons 2, 3, and 4 will be trans, cis, and cis with respect to the  .
.
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the 

If the hydroxyl group attached to carbon 1 ends up trans to the 

The alpha ring structure of D-galactose bonds the carbon 1 hydroxyl group trans to the carbon 5 

← Didn't Know|Knew It →
Which of the following ring structures represents the anomeric alpha ring structure of D-mannose?

Which of the following ring structures represents the anomeric alpha ring structure of D-mannose?

Tap to reveal answer
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the  attached to carbon 5, while those that are in the left position end up cis to the
attached to carbon 5, while those that are in the left position end up cis to the  attached to carbon 5.
attached to carbon 5.
If the hydroxyl group attached to carbon 1 ends up trans to the  attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the
attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the  attached to carbon 5, the ring structure is considered beta.
attached to carbon 5, the ring structure is considered beta.
The alpha ring structure of D-mannose bonds the carbon 1 hydroxyl group trans to the carbon 5  group. The hyroxyl groups on carbons 2, 3, and 4 will be cis, cis, and trans with respect to the
group. The hyroxyl groups on carbons 2, 3, and 4 will be cis, cis, and trans with respect to the  .
.
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the 

If the hydroxyl group attached to carbon 1 ends up trans to the 

The alpha ring structure of D-mannose bonds the carbon 1 hydroxyl group trans to the carbon 5 

← Didn't Know|Knew It →
Identify the aldose pictured, including its alpha or beta designation.

Identify the aldose pictured, including its alpha or beta designation.

Tap to reveal answer
The structure pictured is mannose because the hydroxyl groups at carbons 2, 3, and 4 are situated cis, cis, and trans (respectively) to the  attached to carbon 5.
attached to carbon 5.
The mannose pictured is in alpha form because the hydroxyl group at carbon 1 is trans to the  attached to carbon 5.
attached to carbon 5.
The structure pictured is mannose because the hydroxyl groups at carbons 2, 3, and 4 are situated cis, cis, and trans (respectively) to the 
The mannose pictured is in alpha form because the hydroxyl group at carbon 1 is trans to the 
← Didn't Know|Knew It →
Which of the following correctly describes a reducing sugar?
Which of the following correctly describes a reducing sugar?
Tap to reveal answer
A reducing sugar contains a hemiacetal/hemiketal group which means that in its open chain form it contains a ketone/aldehyde group. Sugars containing a free aldehyde group can be oxidized to a carboxylic acid, while sugars containing a free ketone group must be tautomerized to an aldehyde group through an ene-diol intermediate (shown below), and this can be oxidized to a carboxylic acid.

Reducing sugars are detectable with the formation of either a precipitate or a solution color change after addition of Tollens' Reagent, Benedict's solution, or Fehling's solution.
A reducing sugar contains a hemiacetal/hemiketal group which means that in its open chain form it contains a ketone/aldehyde group. Sugars containing a free aldehyde group can be oxidized to a carboxylic acid, while sugars containing a free ketone group must be tautomerized to an aldehyde group through an ene-diol intermediate (shown below), and this can be oxidized to a carboxylic acid.

Reducing sugars are detectable with the formation of either a precipitate or a solution color change after addition of Tollens' Reagent, Benedict's solution, or Fehling's solution.
← Didn't Know|Knew It →
Alpha-D-glucopyranose:

Glucose (pictured) is defined as which of the following?
Alpha-D-glucopyranose:

Glucose (pictured) is defined as which of the following?
Tap to reveal answer
Glucose in its open chain form (shown below), has a free aldehyde group (an aldose), and it contains six carbons (a hexose). Together, glucose is shown to be an aldohexose.

Glucose in its open chain form (shown below), has a free aldehyde group (an aldose), and it contains six carbons (a hexose). Together, glucose is shown to be an aldohexose.

← Didn't Know|Knew It →
Alpha-D-glucopyranose

Which of the labelled carbon atoms is the anomeric carbon?
Alpha-D-glucopyranose

Which of the labelled carbon atoms is the anomeric carbon?
Tap to reveal answer
The anomeric carbon is formed from the original carbonyl carbon in the straight-chain form of the molecule being attacked by a hydroxyl group to form a hemiacetal. This is seen as a carbon that is bonded to two oxygen atoms. In the case of glucose, the carbon labelled A is a hemiacetal, and is considered to be the anomeric carbon.
The anomeric carbon is formed from the original carbonyl carbon in the straight-chain form of the molecule being attacked by a hydroxyl group to form a hemiacetal. This is seen as a carbon that is bonded to two oxygen atoms. In the case of glucose, the carbon labelled A is a hemiacetal, and is considered to be the anomeric carbon.
← Didn't Know|Knew It →
Shown below is the structure of a sugar molecule.

Which of the following is the most appropriate classification of this sugar?
Shown below is the structure of a sugar molecule.

Which of the following is the most appropriate classification of this sugar?
Tap to reveal answer
In this question, we're given the structure of a sugar molecule, and we're asked to identify which answer choice represents the correct identification of this molecule.
To answer this question, there are two things we need to look at. For one, we need to determine whether it is an aldehyde sugar (aldose) or a ketone sugar (ketose). The first carbon atom in the molecule (shown at the very top in the image) is shown as  . This means that the carbon contains a double bond to the oxygen. Furthermore, since it also contains a bond to a hydrogen, we can conclude that this is an aldehyde functional group. Consequently, we know that this must be an aldose sugar.
. This means that the carbon contains a double bond to the oxygen. Furthermore, since it also contains a bond to a hydrogen, we can conclude that this is an aldehyde functional group. Consequently, we know that this must be an aldose sugar.
Next, we need to look at the number of carbon atoms in the molecule. In the image shown in the question, there are a total of seven carbon atoms. Thus, this sugar would be classified as a heptose sugar (seven carbons) rather than a hexose sugar (six carbons).
Putting these two pieces of information together, we know that the sugar is both an aldose and a heptose. Therefore, this sugar is an aldoheptose.
In this question, we're given the structure of a sugar molecule, and we're asked to identify which answer choice represents the correct identification of this molecule.
To answer this question, there are two things we need to look at. For one, we need to determine whether it is an aldehyde sugar (aldose) or a ketone sugar (ketose). The first carbon atom in the molecule (shown at the very top in the image) is shown as 
Next, we need to look at the number of carbon atoms in the molecule. In the image shown in the question, there are a total of seven carbon atoms. Thus, this sugar would be classified as a heptose sugar (seven carbons) rather than a hexose sugar (six carbons).
Putting these two pieces of information together, we know that the sugar is both an aldose and a heptose. Therefore, this sugar is an aldoheptose.
← Didn't Know|Knew It →
Which of the following lipids is polyunsaturated?
I. (9Z,12Z,15Z)-octadeca-9,12,15-trienoic acid
II. Octadecanoic acid
III. (9Z,12Z)-octadeca-9,12-dienoic acid
IV. (9Z)-9-Octadecenoic acid
Which of the following lipids is polyunsaturated?
I. (9Z,12Z,15Z)-octadeca-9,12,15-trienoic acid
II. Octadecanoic acid
III. (9Z,12Z)-octadeca-9,12-dienoic acid
IV. (9Z)-9-Octadecenoic acid
Tap to reveal answer
This question tests your knowledge of what an unsaturated lipid is, as well as your ability to obtain structural information from IUPAC names.
Firstly, a polyunsaturated lipid is a long carboxylic acid hydrocarbon chain that features multiple unsaturations, or double bonds. Remember from nomenclature that the stem "-en" indicates double bonds, or alkenes, while "-an" indicates alkanes, or fully saturated hydrocarbons. The relevant stems in the answer choices are italicized below:
I. (9Z,12Z,15Z)-octadeca-9,12,15-_trien_oic acid
II. Octadec_an_oic acid
III. (9Z,12Z)-octadeca-9,12-_dien_oic acid
IV. (9Z)-9-Octadec_en_oic acid
As you can see, "trien" and "dien" in I and III indicate these lipids are polyunsaturated, while "an" in II indicates a fully saturated lipid and "en" in IV indicates a monounsaturation. The lipids are drawn below as well, with the alkenes in polyunsaturated lipids circled in red, and the alkene in the monounsaturated lipid circled in green.

This question tests your knowledge of what an unsaturated lipid is, as well as your ability to obtain structural information from IUPAC names.
Firstly, a polyunsaturated lipid is a long carboxylic acid hydrocarbon chain that features multiple unsaturations, or double bonds. Remember from nomenclature that the stem "-en" indicates double bonds, or alkenes, while "-an" indicates alkanes, or fully saturated hydrocarbons. The relevant stems in the answer choices are italicized below:
I. (9Z,12Z,15Z)-octadeca-9,12,15-_trien_oic acid
II. Octadec_an_oic acid
III. (9Z,12Z)-octadeca-9,12-_dien_oic acid
IV. (9Z)-9-Octadec_en_oic acid
As you can see, "trien" and "dien" in I and III indicate these lipids are polyunsaturated, while "an" in II indicates a fully saturated lipid and "en" in IV indicates a monounsaturation. The lipids are drawn below as well, with the alkenes in polyunsaturated lipids circled in red, and the alkene in the monounsaturated lipid circled in green.

← Didn't Know|Knew It →

Which of these two fatty acids has the lower melting point and why?

Which of these two fatty acids has the lower melting point and why?
Tap to reveal answer
Fatty acids that contain a higher degree of unsaturation (more alkene bonds) will introduce more "kinks" into the hydrocarbon chain. This "kinked" chain does not stack nicely with other fatty acids of its kind and therefore are more likely to slip past each other at lower temperatures. This is primarily due to the van der Waals forces within the unsaturated fatty acids being disrupted with the introduction of double bonds. As a result, unsaturated fatty acids generally have a lower melting point than saturated fatty acids.
Fatty acids that contain a higher degree of unsaturation (more alkene bonds) will introduce more "kinks" into the hydrocarbon chain. This "kinked" chain does not stack nicely with other fatty acids of its kind and therefore are more likely to slip past each other at lower temperatures. This is primarily due to the van der Waals forces within the unsaturated fatty acids being disrupted with the introduction of double bonds. As a result, unsaturated fatty acids generally have a lower melting point than saturated fatty acids.
← Didn't Know|Knew It →
Which of the following is an example of a basic amino acid?
Which of the following is an example of a basic amino acid?
Tap to reveal answer
Lysine's R-group includes an amine (-NH2), which can be ionized by picking up a hydrogen. This ability classifies lysine as basic.
Lysine's R-group includes an amine (-NH2), which can be ionized by picking up a hydrogen. This ability classifies lysine as basic.
← Didn't Know|Knew It →
Which of the following amino acids can participate in the formation of a disulfide bridge?
Which of the following amino acids can participate in the formation of a disulfide bridge?
Tap to reveal answer
Cysteine's R-group contains a sulfhydryl group (-SH), which can participate in the formation of a disulfide bridge in a protein's tertiary and/or quaternary structure. Cysteine is the only amino acid to contain a sulfur atom.
Cysteine's R-group contains a sulfhydryl group (-SH), which can participate in the formation of a disulfide bridge in a protein's tertiary and/or quaternary structure. Cysteine is the only amino acid to contain a sulfur atom.
← Didn't Know|Knew It →
How many essential amino acids are there?
How many essential amino acids are there?
Tap to reveal answer
The nine essential amino acids are valine, leucine, isoleucine, histidine, phenylalanine, tryptophan, methionine, threonine, and lysine. These amino acids must be consumed in the diet, since they cannot be synthesized by adult humans.
The nine essential amino acids are valine, leucine, isoleucine, histidine, phenylalanine, tryptophan, methionine, threonine, and lysine. These amino acids must be consumed in the diet, since they cannot be synthesized by adult humans.
← Didn't Know|Knew It →
Which of the following amino acids is the only one to contain a side chain that results in an achiral amino acid?
Which of the following amino acids is the only one to contain a side chain that results in an achiral amino acid?
Tap to reveal answer
The only achiral amino acid is Glycene. Glycene's side chain is simply a hydrogen. Because a hydrogen already exists on the fundamental structure of an amino acid backbone, a side chain of a single hydrogen atom causes glycene to be achiral.
The only achiral amino acid is Glycene. Glycene's side chain is simply a hydrogen. Because a hydrogen already exists on the fundamental structure of an amino acid backbone, a side chain of a single hydrogen atom causes glycene to be achiral.
← Didn't Know|Knew It →
What is the D/L configuration and the absolute (R/S) configuration of the following amino acid (cysteine)?

What is the D/L configuration and the absolute (R/S) configuration of the following amino acid (cysteine)?

Tap to reveal answer
Cysteine is an unusual amino acid. Although each of the normal biological amino acids have the L configuration (with the exception of glycine, which is achiral), meaning they can be drawn with the Fischer projection as shown:

the fact that cysteine contains a sulfur group makes the side chain a higher priority than the carboxylic acid. Therefore, the absolute configuration of cysteine will be R, and not S like the other amino acids.
Cysteine is an unusual amino acid. Although each of the normal biological amino acids have the L configuration (with the exception of glycine, which is achiral), meaning they can be drawn with the Fischer projection as shown:

the fact that cysteine contains a sulfur group makes the side chain a higher priority than the carboxylic acid. Therefore, the absolute configuration of cysteine will be R, and not S like the other amino acids.
← Didn't Know|Knew It →
Consider L-glutamate, which is shown below.

Using the pKa information provided, what is the isoelectric point (pI) of the given molecule?
Consider L-glutamate, which is shown below.

Using the pKa information provided, what is the isoelectric point (pI) of the given molecule?
Tap to reveal answer
The isoelectric point is the average of the two pKa values around which the molecule has an overall neutral charge. In the case of glutamate, the amino acid is in its neutral (zwitterion) form in between the lower two pKa values. Taking the average of the two values gives the pI.

The isoelectric point is the average of the two pKa values around which the molecule has an overall neutral charge. In the case of glutamate, the amino acid is in its neutral (zwitterion) form in between the lower two pKa values. Taking the average of the two values gives the pI.
← Didn't Know|Knew It →
Consider the amino acid L-lysine, shown below.

Given the pKa values shown, calculate the pI of lysine.
Consider the amino acid L-lysine, shown below.

Given the pKa values shown, calculate the pI of lysine.
Tap to reveal answer
The isoelectric point is the average of the two pKa values around which the molecule has an overall neutral charge. In the case of lysine, the amino acid is in its neutral (zwitterion) form in between the higher two pKa values. Taking the average of the two values gives the pI.

The isoelectric point is the average of the two pKa values around which the molecule has an overall neutral charge. In the case of lysine, the amino acid is in its neutral (zwitterion) form in between the higher two pKa values. Taking the average of the two values gives the pI.
← Didn't Know|Knew It →
Treatment of a peptide with Edman's reagent yielded the following phenylthiohydantoin (PTH) derivative:

What is the side chain of the amino acid at the N-terminus of the peptide?
Treatment of a peptide with Edman's reagent yielded the following phenylthiohydantoin (PTH) derivative:

What is the side chain of the amino acid at the N-terminus of the peptide?
Tap to reveal answer
Treatment of a peptide with Edman's reagent ( ) will remove the N-terminal amino acid. The PTH derivative will look as follows:
) will remove the N-terminal amino acid. The PTH derivative will look as follows:

In the molecule in the question, a sec-butyl replaces the R group. This is the amino acid isoleucine.
Treatment of a peptide with Edman's reagent (

In the molecule in the question, a sec-butyl replaces the R group. This is the amino acid isoleucine.
← Didn't Know|Knew It →
What is the amino acid that is synthesized from the malonic ester synthesis shown?

What is the amino acid that is synthesized from the malonic ester synthesis shown?

Tap to reveal answer
First step: Deprotonation of alpha hydrogen and formation of the malonic ester enolate.
Second step: bimolecular nucleophilic substitution, resulting in addition of the isopropyl group to the alpha carbon.
Third step: hydrolysis of amide and ester bonds, followed by decarboxylation
The synthesis forms valine, because the alkyl halide that is attacked is an isopropyl group (Valine's side chain is an isopropyl group). A more detailed mechanism is shown below.

First step: Deprotonation of alpha hydrogen and formation of the malonic ester enolate.
Second step: bimolecular nucleophilic substitution, resulting in addition of the isopropyl group to the alpha carbon.
Third step: hydrolysis of amide and ester bonds, followed by decarboxylation
The synthesis forms valine, because the alkyl halide that is attacked is an isopropyl group (Valine's side chain is an isopropyl group). A more detailed mechanism is shown below.

← Didn't Know|Knew It →